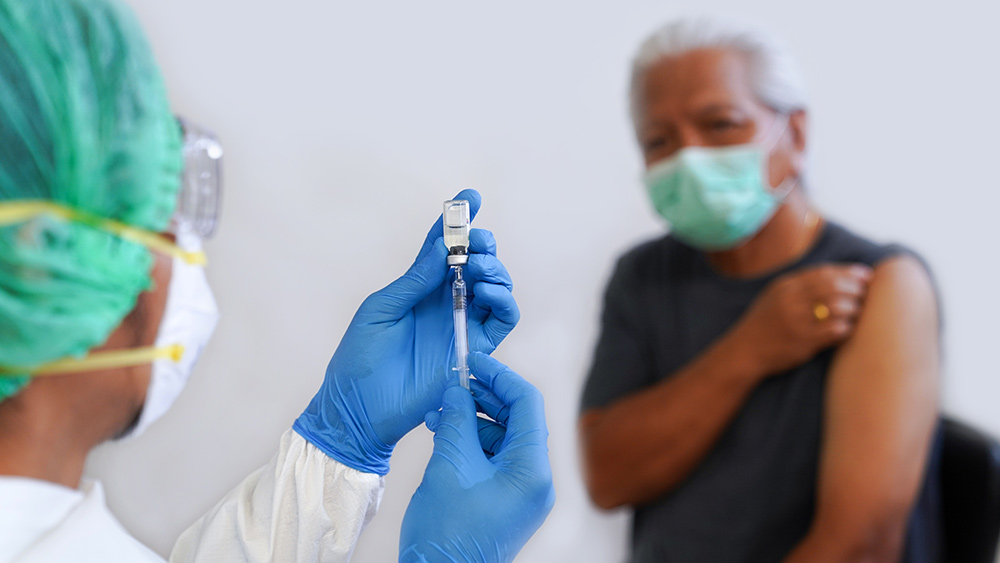
Chinese pharmaceutical firm Sinovac gets $500 million, boosting its coronavirus vaccine production
12/30/2020 / By Ramon Tomey

Chinese drugmaker Sinovac Biotech has obtained a cash investment to help boost production of its COVID-19 vaccine. Generic drugs firm Sino Biopharmaceuticals announced on Dec. 7 that it acquired a 15 percent stake in Sinovac Life Sciences, the subsidiary in charge of vaccine manufacturing. Sinovac expects a yearly production output of 300 million Wuhan coronavirus vaccine doses, but this capacity will double once a second production facility is constructed by the end of 2020.
Sinovac chief Yin Weidong said the investment would enable the company “to improve [its] vaccine sales capabilities [and] expand in Asia markets.” Meanwhile, Sino Biopharmaceuticals said in an exchange filing that “the capital contribution will introduce a key strategic partner to the group.” It also said that “both parties will join efforts … in further improving vaccine sales capability, [expanding] to overseas markets … [and developing] new technologies.”
The investment’s timing comes as Sinovac looks to increase production and distribution of its COVID-19 vaccine candidate following phase three trials. As of writing, the Sinovac jab is undergoing large-scale trials in different countries such as Brazil, Chile, Pakistan and Indonesia. Sinovac’s COVID-19 jab joins two other candidates by the state-run Sinopharm as possible vaccines for worldwide distribution.
Almost a million Chinese citizens have been immunized using the Sinopharm jabs through an emergency program commenced in July. Outside China, the North African nation of Morocco has expressed interest in the Sinopharm COVID-19 vaccines for its own immunization program to start this month.
Meanwhile, Western vaccine candidates have also shown promise in clinical trials. The mRNA coronavirus vaccines from Pfizer/BioNTech and Moderna have demonstrated efficacy rates above 90 percent in clinical trials. However, they need to be transported and stored at sub-zero temperatures: The Pfizer/BioNTech jab needs to be in a consistent -70 degrees Celsius environment, while the Moderna jab needs to be kept at -20 degrees Celsius for it to work. Sinovac and Sinopharm’s inactivated vaccines can be distributed more easily as they do not require cold temperatures.
Sinovac’s coronavirus jab is being tested in different countries such as Indonesia and Brazil
Indonesian state news agency Antara reported that 1.2 million doses of Sinovac’s COVID-19 jab were delivered to the capital Jakarta Dec. 6. Indonesian President Joko Widodo said: “We are still working on [another] 1.8 million doses … [to] arrive in early January 2021,” adding that the country was “very grateful that the vaccine is already available.”
Widodo added that Indonesia’s National Agency of Drug and Food Control was already working on approvals so the vaccine could be distributed. (Related: Indonesia, which has the second-worst COVID-19 outbreak in Asia, is now the testing ground for a Chinese vaccine.)
Meanwhile, Health Minister Terawan Agus Putranto assured reporters during a Dec. 7 virtual press briefing that the Wuhan coronavirus vaccines developed by the Chinese pharmaceutical firm arrived without any defects and ready for delivery to storage warehouses. He remarked that the doses which arrived the previous day had “no defect in packaging, content and temperature during the delivery.”
Putranto added: “Hopefully, the COVID-19 vaccination would be conducted on time and run smoothly.”
The state of Sao Paulo in Brazil also obtained doses from Sinovac. It procured 60 million doses from the Chinese drugmaker in October, with state governor Joao Doria asking Brazilian health regulator Anvisa to register Sinovac’s candidate. It subsequently gave permission to proceed with large-scale trials in the state.
Brazilian Health Minister Eduardo Pazuello also announced in the same month that the federal government would purchase 46 million doses from Sinovac.
However, the purchase from Sinovac was met with opposition from both government officials and ordinary citizens. Brazilian President Jair Bolsonaro said the country would not buy the vaccine and advised Pazuello to “find someone else willing” to purchase the doses. A number of residents gathered at downtown Sao Paulo to protest against any mandatory vaccination, with some protesters calling for Doria’s resignation. (Related: “We’re not guinea pigs!” – Brazilians push back against mandatory coronavirus vaccinations.)
Data collated by Johns Hopkins University shows that Brazil has the third highest worldwide COVID-19 caseload at 6.6 million, with 5.8 million recoveries and 177,317 deaths. Meanwhile, Indonesia has 581,550 COVID-19 cases with 479,202 recoveries and 17,867 deaths.
Visit Vaccines.news to find out the latest about vaccines being developed to fight the ongoing pandemic.
Sources include:
Submit a correction >>
Tagged Under:
Brazil, cash boost, cash investment, Chinese pharmaceuticals, chinese vaccines, Clinical trials, company acquisition, coronavirus vaccine, covid-19 pandemic, efficacy rates, Indonesia, mandatory vaccination, Medical Tyranny, minority shareholder, Morocco, phase three trials, Sino Biopharmaceuticals, Sinopharm, Sinovac Biotech, vaccination, vaccination mandates, vaccine candidate, vaccine efficacy, vaccine trials, vaccines, Wuhan coronavirus
This article may contain statements that reflect the opinion of the author
RECENT NEWS & ARTICLES
Vaccines.News is a fact-based public education website published by Vaccines News Features, LLC.
All content copyright © 2018 by Vaccines News Features, LLC.
Contact Us with Tips or Corrections
All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.